Pharmacokinetics and its Applicability – Dr. Sandeep Kaushal
Pharmacology is the study of the interaction of drugs with biological systems. This includes research on the chemical makeup, biological processes, physiological and behavioral impacts, mechanisms of action, and both therapeutic and non-therapeutic applications of pharmaceuticals.
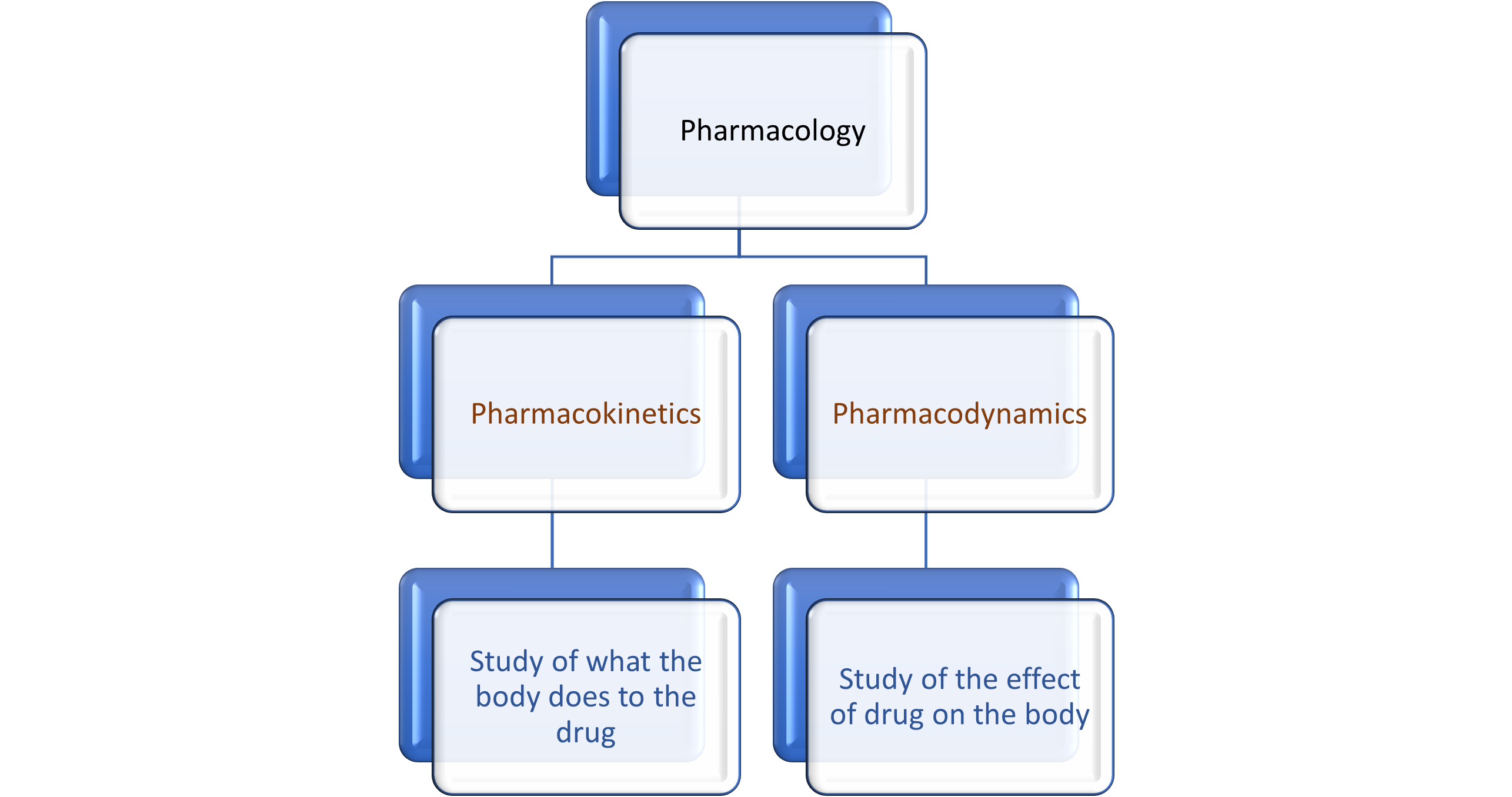
Pharmacology is broadly categorized into two terms:
PHARMACOKINETICS IN PHARMACOLOGY
The term ‘Pharmacokinetics’ is derived from ancient Greek words, pharmakon meaning ‘drug’ and kinetikos meaning ‘moving’ or put in motion. Pharmacokinetics is a journey of a drug through the body, and in this, a drug passes through four different phases- Absorption, Distribution, Metabolism, and Excretion, which are called ADME properties.
The selection and modification of drug-dose schedules are aided by pharmacokinetic parameters.
Let’s discuss the pharmacokinetic parameters:
- ABSORPTION:
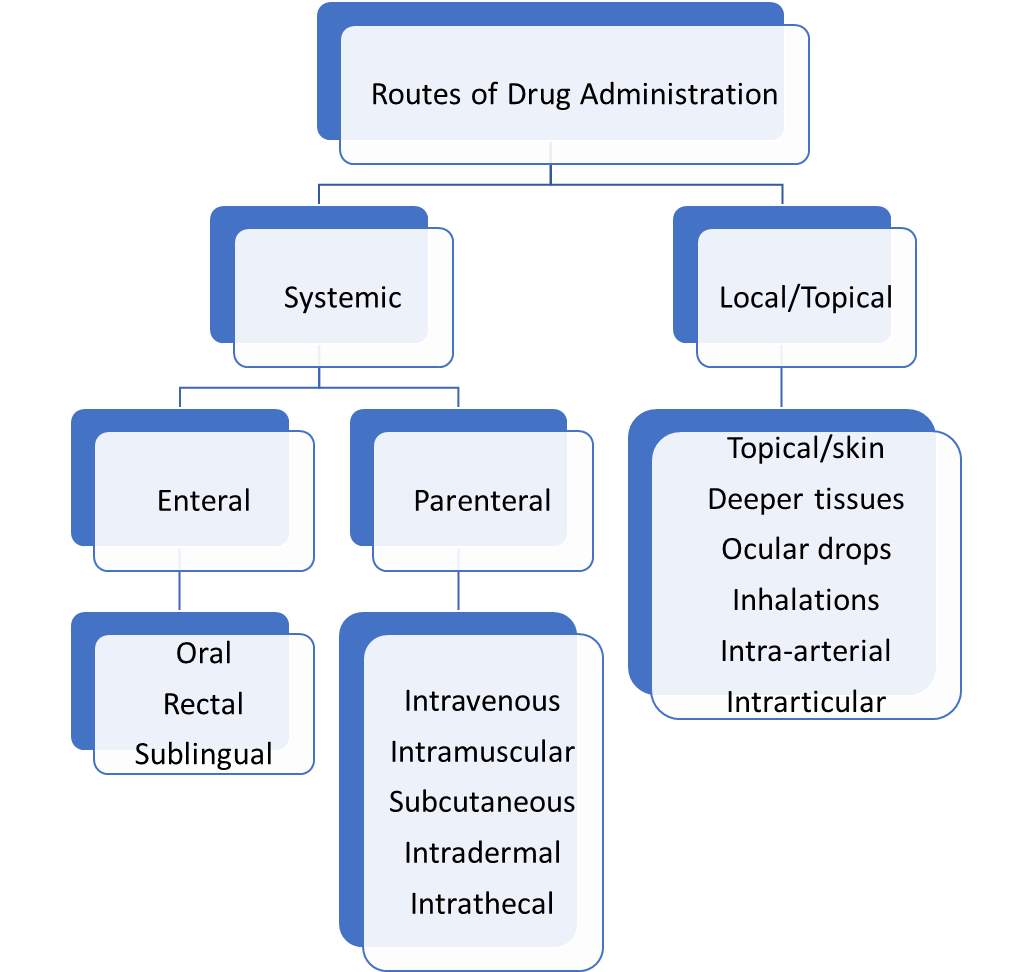
Absorption is the movement of a drug to the site of action from the site of administration. Various factors affect the extent and rate of absorption of the drug, such as routes of drug administration, dosage form, physiochemical properties of the drug, bioavailability, pharmacogenetic factors, etc.
First Pass Metabolism: It is a phenomenon in which a drug is metabolized or transformed at a specific site before the drug reaches its site of action or systemic circulation, resulting in a lower concentration of the active substance. Examples of the drugs that undergo this phenomenon in the liver are propranolol, lidocaine, and clomethiazole; in the gut wall are sex hormones.
In the case of intravenous or intra-arterial administration, the drug bypasses the first pass metabolism and enters the circulation directly.
Watch this free video of one of the best online pharmacology courses in MBBS
Drug distribution is the process by which a drug travels from the administered site after absorption via the blood vessel walls to the sites of action.
The drug is distributed via various body fluid compartments, such as plasma, interstitial fluid compartments, and trans-cellular compartments.
Factors affecting the distribution rate of the drug:
- Protein binding of drug
- Plasma concentration of drug (Cp)
- Clearance
- Physiological barriers to distribution
- Drug affinity of drugs to certain tissues
3. METABOLISM:
Drug metabolism, also known as biotransformation, is the process by which the body metabolizes drug molecules. The principal site of drug metabolism is the liver.
Enzymes responsible for drug metabolism are classified into two categories:
- Microsomal enzymes: They are present in the smooth endoplasmic reticulum of the liver, kidney, and GIT. Example: cytochrome P450.
- Non-microsomal enzymes: They are present in the cytoplasm, and mitochondria of different organs. Examples: esterase, amidase, and hydrolase.
Two Phases of Metabolism: Phase I metabolism converts a parent drug to polar active metabolites, via., oxidation, reduction, and hydrolysis reactions, while phase II metabolism converts a parent drug to polar inactive metabolites including conjugation reactions.
Watch this video of Pharmacology for UnderGrads and gain an in-depth understanding of Pharmacology in MBBS.
Drug excretion refers to the removal of drugs in unchanged or modified form out of the body.
The major route of excretion:
- Renal excretion: It involves three major physiological processes, namely, glomerular filtration, active tubular secretion, and passive tubular reabsorption.
- Hepatobiliary excretion: The drug with a molecular weight of more than 300 Da and polar drugs are excreted in the bile.
- Gastrointestinal excretion: After the oral administration of a drug, a part of it is not absorbed and excreted in the feces.
- Pulmonary excretion: Volatile drugs, such as gaseous anesthetics, are excreted via the lungs into expired air.
Saliva, sweat, tears, breast milk, vaginal fluid, nails, and hair are considered the minor routes of drug excretion.
VARIOUS PHARMACOKINETIC PARAMETERS:
- Half-life: It is the time taken for the drug concentration in blood or plasma to reduce to half of the original amount, i.e. the amount of the drug in the body is reduced by 50%. A drug’s half-life varies from patient to patient because of a variety of patient and drug-specific factors.
The formula for the half-life is t½ = 0.693 × Vd /CL where,
Vd is the volume of distribution and CL is a clearance factor.
2. Order of Kinetics: For many medications, the most typical is First order kinetics, meaning that a fixed proportion of the drug is eliminated from the body at regular intervals of time. Only a few medications, like ethanol and phenytoin, undergo zero order kinetics wherein, a fixed quantity of the medicine is eliminated after each period.
3. Clearance of a Drug: It is the volume of plasma cleared or removed out of the drug by hepatic and/or renal excretion, among other organs.
Clearance = Rate of Elimination
Plasma drug concentration
And, Total clearance is calculated by Clt = Clh + Clr + Clothers where,
Clt = Total clearance
Clh = Hepatic clearance
Clr = Renal clearance
Clother = Clearance from all other routes
4. Steady-state plasma concentration: When a drug dose is administered regularly over a certain time, a steady state is reached. It is a point where the amount of medication absorbed and the amount removed from the body is in equilibrium. For instance, a medication that has a half-life of 6 hours will likely reach a steady state after being administered for more than 24 hours (More than 4 half-life).
To access online pharmacokinetics lecture notes, download the DigiNerve app.
Dr. Sandeep Kaushal
MD, MAMS, FCP(ACCP), FIMSA, MBPhS, ACME
Dean Academics, Professor, and Head,
Department of Pharmacology,
Dayanand Medical College and Hospital,
Ludhiana, Punjab
Related post